
Table 7 - Semi-qualitative 5x5 risk evaluation matrix. Table 7 shows the risk evaluation and risk acceptability for Palpreast. For each identified hazardous situation, the manufacturer decides if risk reduction is required, on the basis of its acceptability criteria defined in the risk management plan. Table 6 - Semi-quantitative 5x5 risk matrix for Palpreast Evaluation RiskĪccording to ISO 14971, the “Evaluation of Risk” is defined as the “Process of comparing estimated risk against given risk criteria to determine acceptability of the risk”. Table 4 - Semi-qualitative probability levels Table 3 - Five qualitative severity levels
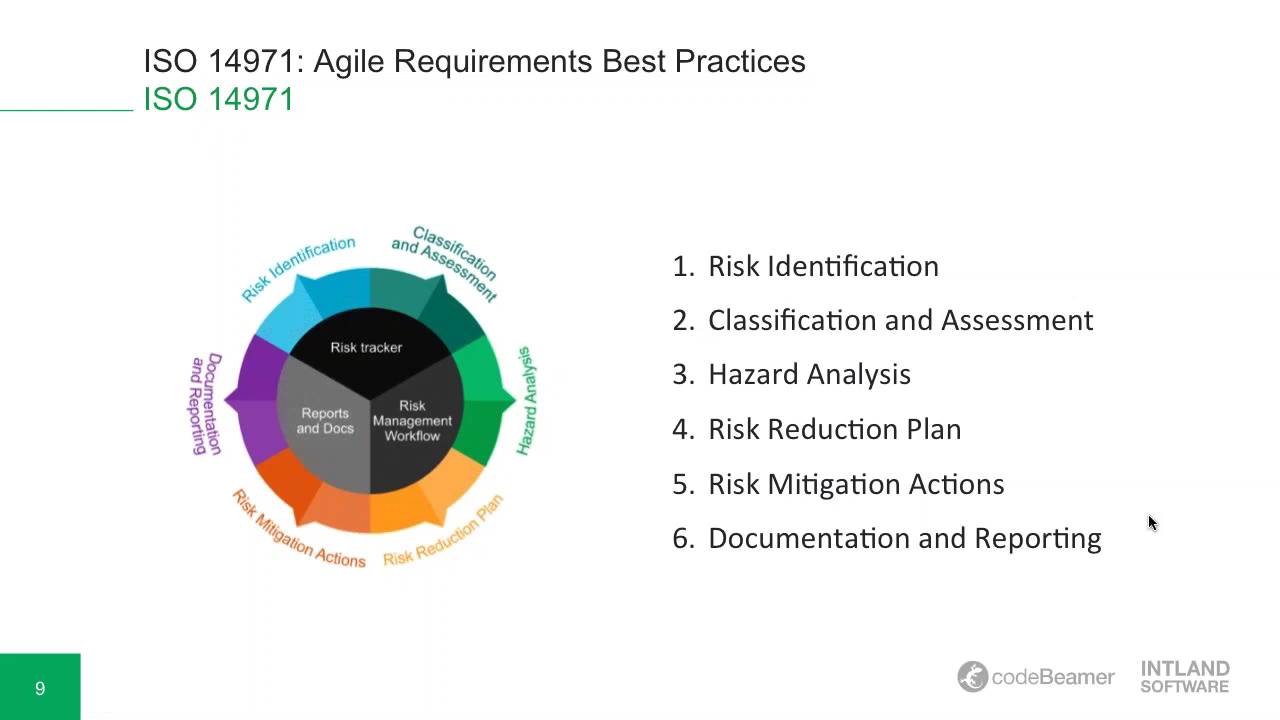
Handouts are hazard analysis forms and HA report template.This section was developed step-by-step, to be used also a teaching material and as template for other projects developed through UBORA.Ī complete risk analysis has been performed taking into consideration Palpreast device. We will explain these concepts and provide examples so that you can expertly perform the process. Hazard Analysis, described in ISO 14971, is the most powerful of the risk management tools, but it is very confusing. Risk Analysis is required in a FDA product submission.įDA recommends using ISO 14971 as a guide and has accepted it as a recognized standard. Requirements for each step of the risk management process, including the risk management plan, development of risk acceptability criteria, risk analysis, risk evaluation, risk control, risk/ benefit analysis, postproduction analysis will be explained.įDA expects that as part of a product development program risk management will be conducted and risks will be mitigated as far as practical.
RISK ANALYSIS ISO 14971 TEMPLATE SOFTWARE
Risk level can determine the extent of CAPA investigations, validations effort, etc.Īpplication of ISO 14971 principles to software risk management will be explained
RISK ANALYSIS ISO 14971 TEMPLATE HOW TO
We will explain how to integrate Human Factors studies into the Hazard Analysis and how to integrate Hazard Analysis into the design program. How to deal with residual risk will be discussed. Examples of hazards and hazardous situations will be discussed. We will go step by step through a template for risk analysis so that the process is clear. The confusing terms "hazard", hazardous situation", "harm", "causative event", "ALARP", "risk index", "benefit/ risk ratio", and "residual risk" will be explained. In this webinar we will explain in detail the process of conducting a hazard analysis.

FMEA and FTA consider only fault conditions and are more suited as reliability tools than as product safety tools. This is the most powerful of the risk management techniques because it considers risks in normal operation as well as fault conditions. Hazard Analysis is described in ISO 14971. FDA also expects that a post production risk management program be implemented.įDA recommends using ISO 14971 as a guide and has accepted it as a recognized standard. The US FDA expects that as part of a product development Design Control Program risk management will be conducted.
